
Home, Auto Repair Library, Auto Parts, Accessories, Tools, Manuals & Books, Car BLOG, Links, Index



There have always been alternatives to gasoline, but except for the artificial fuel shortages created by the OPEC oil embargo in the 1970s which fueled the first so-called "energy crisis," alternative fuels have mostly remained on the sidelines. Well now they are being called up to become major sources of energy as oil prices soar once again.
Propane and natural gas have traditionally been the alternative fuels of choice for fleet vehicles. Until recently, the primary motivation for converting to alternative fuels was to reduce operating and maintenance costs.
On a cents per mile basis, natural gas is probably the cheapest alternative fuel, followed by propane, then methanol alcohol and finally ethanol alcohol.
The up front costs for converting a vehicle to propane range from $1500 to $3000, and $2500 to $4000 for compressed natural gas depending on the application. This only includes the cost of modifying the fuel and ignition system, and does not include any costs associated with "optimizing" the engine through internal modifications (which we'll get to later). Despite the high initial conversion costs, most fleets get a fairly rapid payback thanks to the savings realized by lower fuel and operating costs.
Alternative fuels have worked for fleets because fleets can set up their own centralized refueling facilities. But the availability of refueling locations has been and will continue to be an obstacle to the widespread acceptance of any alternative to gasoline. It costs big bucks to build duplicate refueling and distribution facilities for alternative fuels. So we've been faced with the "chicken and egg" syndrome. The car makers do not want to build vehicles for the general public that can only run on a special alternative fuel because the fuel is not readily available. And fuel suppliers do not want to add alternative fuel infrastructure if there are few vehicles that can use an alternative fuel.
E85 (85 percent ethanol alcohol and 15% gasoline) and M85 (85% methanol alcohol and 15% gasoline) currently are the only alternative fuels that have some availability (though limited) because they can be burned in specially-equipped "flex-fuel" vehicles. Flex-fuel vehicles can run on anything from straight gasoline up to 85% ethanol or methanol alcohol. See Methanol and Ethanol Alcohol subheads below.

Unlike E85 and M85 which are both liquid fuels, propane (also known as Liquefied Petroleum Gas, LP-Gas or LPG), and natural gas are both gaseous fuels. That means they are a vapor at room temperature and must be contained in a special high pressure fuel cylinder. Natural gas can be used as compressed natural gas (CNG) or Liquefied Natural Gas (LNG).
About 60% of the propane that's produced comes from natural gas wells. The rest is a byproduct of crude oil refining. The price of propane fluctuates seasonally and like all energy sources has been rising in price.
Propane has been used since the 1930s, so it has a long history as a motor vehicle fuel. Today it is used to power cabs, school buses, recreational vehicles, delivery trucks, farm vehicles and zillions of industrial fork lift trucks. The National Propane Gas Association estimates there are half a million propane-powered vehicles on the road in the U.S today. Most burn propane exclusively, but some have dual-fuel capability and can switch back and forth with gasoline. NLPGA also says there are about 10,000 LP-gas refueling facilities nationwide, which means the fuel is available in many areas.
As a fuel, propane contains somewhat less heat energy per gallon than gasoline (91,547 BTUs versus 116,000 BTUs for gasoline). But on a pound per pound basis, it delivers almost the same energy as gasoline (21,591 BTUs) -- which means an engine converted to propane consumes about 10% more fuel in terms of miles per gallon with little or no loss in horsepower. Carbon monoxide (CO) emissions are naturally low but hydrocarbon (HC) emissions may actually be somewhat higher with propane. Even so, the emissions are considered to be "less reactive" in forming smog.
Propane produces less carbon and blowby in the engine itself, which extends the life of the spark plugs and oil. Oil change intervals can usually be doubled or even tripled with propane.
Propane is a gaseous fuel and boils at -44 degrees F., so it must be kept in a pressurized fuel tank. Pressurizing propane turns it into a liquid, which allows more fuel to be stored in a smaller volume. A typical 25 gallon capacity propane tank holds about 180 lbs. of liquid fuel at 175 psi when filled to 80% capacity -- which gives approximately the same driving range as an equivalent tank of gasoline.
Propane's octane rating is 103 which allows it to handle more compression that gasoline. Propane is also a "dry" fuel which means it enters the engine as a vapor rather than as droplets of liquid. Dry fuels mix with air better than wet fuels, and provide a more uniform air/fuel mixture to each of the engine's cylinders. This promotes cleaner combustion, easier starting and fewer cold driveability problems.
But dry fuels such as propane and natural gas don't cool or lubricate the valves like gasoline does, which means valve burning, wear and recession can be a problem if the engine doesn't have hard seats. Any engine that's designed to burn unleaded gasoline can handle propane without problems, but older engines or truck engines may have to have hard seats installed.
If an engine is being rebuilt for a vehicle that burns propane, the stock compression ratio can be increased up to 10:1 to take advantage of propane's higher octane rating. Special attention also needs to be paid to the valves. Hard seats must be installed if the heads don't already have them. Most experts recommend using a 1/16 to 3/32 in. wide seat on the intakes and a full 3/32 in wide seat on the exhaust valves with no interference angle. Both valve and seats should be cut or ground to 45 degrees. Careful attention should be paid to valve and seat concentricity to prevent hot spots.
Because propane is a dry fuel, it doesn't "wash down" the cylinder walls like gasoline does. This reduces cylinder bore wear but can also create ring seating problems if the wrong type of rings are used or the bores haven't been honed properly.
Recommendations vary, but one leading ring manufacturer says to use either cast iron or moly top compression rings in propane fueled engines. Chrome rings are not recommended. The cylinder bores should be bored to within .003 in, of final size, then rough honed with 180 or 220 grit stones to within .0005 in. of final dimensions and finish honed with a 280 grit stone -- or stroked six to 10 times with a 400 grit stone to "plateau" the surface.
Other engine modifications that can be made to optimize performance with propane include eliminating manifold preheating by blocking the exhaust crossover passageway on a V6 or V8 engine, altering the ignition curve (electronically or by recalibrating the distributor) and decreasing the spark plug gap (.035 in. is recommended with electronic ignition).
Another change that's recommended is to use a motor oil specially formulated for LP-gas. Ordinary motor oils contain additives to neutralize the blowby contaminants from gasoline. But these additives aren't needed with propane and can actually cause deposits of their own. Oils designed for propane applications have a "low ash" additive package.

Like propane, natural gas is a dry gaseous fuel. The same recommendations that apply to modifying an engine for propane also apply for natural gas. For a straight conversion or dual-fuel application, switching to natural gas will produce about a 10% loss of horsepower -- unless the engine is optimized for natural gas by increasing compression, in which case there's no difference in power.
Natural gas is primarily methane. As a motor fuel, it has the highest octane rating of any of the other alternatives at 130, which means it can handle compression ratios of up to 15:1! Methane is also the cleanest burning fuel with substantially lower CO and HC emissions than propane, gasoline, or the alcohols. It's also the cheapest of the bunch at 72 cents for the energy equivalent of a gallon of gas.
But methane has some drawbacks. One is it's low energy content. It takes about 100 cubic feet of methane to deliver the same amount of horsepower as a gallon of gasoline. Consequently, it requires heavy high pressure fuel tanks. A typical 140 lb. natural gas fuel tank filled with compressed gas at 2600 to 300 psi holds the energy equivalent of only about four gallons of gasoline -- which limits the driving range to about 90 to 120 miles unless additional tanks are added (which also add weight and bulk to the vehicle).

One way to extend the driving range of a natural gas powered vehicle is use liquefied natural gas (LNG) rather than compressed natural gas. Chilling methane to -260 degrees F. reduces its volume by a factor of 630 to 1, allowing more fuel to be stored in a smaller tank. But a super cold cryogenic fuel tank can't keep the methane liquid indefinitely. As the fuel warms up, it begins to vaporize and must either be vented or used. LNG costs more than CNG because the equipment that's needed to chill the gas is expensive.
Compressing methane isn't cheap either. A two-stage high pressure compressor station for refueling large numbers of vehicles can cost up to $250,000! Smaller compressors for overnight home refueling are available, but even these cost as much as $3000 apiece.
Another drawback that limits methane's potential as an alternative fuel for mass consumption is a limited infrastructure for refueling. Natural gas pipelines are everywhere, and with fracking the supplies of natural gas have expanded significantly causing a drop in natural gas prices. But there are only about 500 refueling facilities for CNG vehicles nationwide.
Natural gas powered vehicles are available from the domestic auto makers. GM and Ford have both produced CNG pickups while Chrysler has built CNG-powered vans. But most of these vehicles are for fleet users, not the general public. Even so, from 2003 to 2011, the number of natural gas-powered vehicles on the road increased at an annual rate of 10%
For information about CNG vehicles you can buy, Click Here.

If you've watched the Indianapolis 500 or an alcohol-fuel dragster, you have seen what methanol can do on the race track. As a high octane racing fuel, it's great. Racers love it because of its high octane rating of 105 allows an engine to run a higher compression ratio and to inhale in a denser fuel charge for up to a 10% increase in horsepower.
But great racing fuels don't necessary make great everyday fuels. Methanol contains only 57,000 BTUs per gallon, which is half that of gasoline. So an engine has to burn nearly twice as much methanol to travel the same distance it would on gasoline.
Methanol is cheaper than gasoline, but when the increased fuel consumption is taken into account the cost per mile for methanol is substantially higher. Even with an optimized engine, methanol delivers only about 60% of the fuel mileage of gasoline limiting a vehicle's driving range unless the size of the fuel tank is nearly doubled.
Most of the methanol that's available today is made from natural gas, but it can also be made from coal -- which is the real reason why methanol is being pushed as a major alternative to gasoline (the U.S has plenty of coal).
The HC, CO and NOX emissions produced by methanol are a little less than those produced by gasoline, but like propane the HC emissions are the lighter molecules that are less reactive. So that's why you hear statements like "methanol is 30 to 50% cleaner than gasoline." But methanol makes some nasty emissions of its own, namely formaldehyde which is 10 times higher than gasoline. A modified catalytic converter is needed to limit these emissions.
Among its other drawbacks, methanol is a very potent solvent, so hoses and plastic components in the fuel system must be made of compatible materials. Methanol also absorbs moisture, which is good, but moisture can cause corrosion problems inside steel tanks and fuel lines requiring the use of stainless steel or specially plated steel.
Fuel availability is another problem with methanol. In California where methanol has been promoted in the past, there are fewer than 100 stations that sell it. Nationwide, methanol outlets are about as scarce as those for compressed natural gas.
Straight methanol may be great on the race track, but it's not so great when used straight on the street -- especially in cold weather. Like gasoline, methanol is a liquid and must first be vaporized before it will combust. But below 40 degrees F. methanol vaporizes very poorly. To overcome cold starting problems, a supplemental gasoline starting system is needed or the fuel must be blended with some gasoline to give it the extra "kick" to get it going. One such blend is called "M85," a mixture of 85% methanol and 15% gasoline.
Ford's "Flex Fuel Vehicle" (FFV) program, GM's "Variable Fuel Vehicle" (VFV) program and Chrysler's "Gasoline Tolerant Methanol Vehicle"(GTMV) program are all based on vehicles that can burn M85 or any other blend of methanol and gasoline. The trick that makes this possible is a fuel sensor that measures the relative proportions of the two fuels in the fuel line so the engine computer can make the necessary adjustments in fuel delivery and spark timing. The flex fuel capability gives consumers the option of burning either fuel or any combination thereof.

 Ethanol (grain) alcohol is yet another alternative. At one time, it rivaled gasoline as the motor fuel of choice. But that was in the early days of the automobile. In the 1970s it resurfaced as a fuel extender in Gasohol (a blend of 10% ethanol and 90% gasoline). Today, it is widely used as an octane boosting additive in many premium and mid-grade gasoline. Most regular and premium grades of gasoline contain 10 percent ethanol (E10), and the EPA has approved the use of 15 percent ethanol (E15) in 2002 and newer vehicles. Ethanol is also used in "reformulated" gasoline because it oxygenates gasoline and helps it burn cleaner with lower HC and CO emissions.
Ethanol (grain) alcohol is yet another alternative. At one time, it rivaled gasoline as the motor fuel of choice. But that was in the early days of the automobile. In the 1970s it resurfaced as a fuel extender in Gasohol (a blend of 10% ethanol and 90% gasoline). Today, it is widely used as an octane boosting additive in many premium and mid-grade gasoline. Most regular and premium grades of gasoline contain 10 percent ethanol (E10), and the EPA has approved the use of 15 percent ethanol (E15) in 2002 and newer vehicles. Ethanol is also used in "reformulated" gasoline because it oxygenates gasoline and helps it burn cleaner with lower HC and CO emissions.
Ethanol is much like methanol as far as its fuel qualities are concerned. It has a high octane rating (102) and can be run with higher compression ratios to increase overall engine performance. Ethanol contains more energy per gallon than methanol (76,000 BTUs) but not as much as gasoline so fuel consumption is about 50 to 60% higher with straight ethanol compared to gasoline. Like methanol, ethanol is also sluggish to vaporize at cold temperatures so it works best when blended with gasoline.
Ethanol's primary attraction as an alternative fuel is that it comes from renewable resources. Ethanol is made by fermenting sugar which can come from almost any crop or even garbage. The higher the sugar content, the more alcohol a crop can produce. Sugar cane and sugar beets are good choices for making ethanol, but in the Midwest, the most common crop (and cheapest to produce) is corn, so corn has become the feedstock of choice for making most of the ethanol in the U.S. The crop is ground, mixed with water and yeast to create "mash." After several days, the fermented liquid is then boiled and distilled to separate out the alcohol, a process that requires energy, adds cost and makes ethanol the most expensive fuel of the major alternatives.
Some critics have said that it takes more energy to produce ethanol alcohol from corn than it yields. The US Dept. of Energy has determined that ethanol from corn yields MUCH MORE energy than required to produce it. The latest findings say it takes 740,000 Btu of fossil energy to create and deliver ethanol containing 1 Billion Btu's. The calculation includes the energy consumed to grow and harvest the corn (tractor fuel), the energy to process and ferment the corn, and distill the alcohol (electricity and natural gas), and the energy to transport and deliver the fuel (truck fuel).
Argonne National Laboratory near Chicago says corn ethanol delivers a very positive energy balance. Ethanol requires 24.3% more overall energy input than gasoline to produce, but consumes 32.9% less fossil energy and expends 69.5% less petroleum energy overall. Turning oil into gasoline and diesel fuel requires transporting crude oil to refineries that are often half way around the world (if the oil comes from the Mid-East to the U.S.), and requires a lot of energy to refine the oil into its various end products. And these calculations do not include the HUGE military costs of protecting overseas oil sources.
Ethanol production has been rising, and dozens of ethanol plants have been built across the corn belt states. The demand for ethanol has driven up corn prices. Corn that used to sell for $2 a bushel increased to $5 to $6 a bushel, but more recently has been around $3.40 a bushel (2018). Higher corn prices are good news for farmers but bad new for ethanol fuel producers because it makes ethanol more expensive to produce.
If you live on a farm and grow your own corn, why not make your own alcohol fuel?

Another book you might find interesting is The Ethanol Papers Book by Marc J. Rauch of The Auto Channel. Marc takes an in-depth no B.S. look at the many issues and myths surrounding the use of ethanol alcohol as a motor fuel. .
As the technology for making ethanol out of non-food sources such as cellulose (cork stalks, wood fiber, even paper) improves, the energy balance favoring ethanol will be even better. Making ethanol out of cellulose requires special enzymes that can break down the starchy fibers into fermentable sugars. But the enzymes are expensive. They used to cost about $5 a gallon and have come down to less than 20 cents a gallon. Analysts say if the price of enzymes can get down to ten cents a gallon, making ethanol from cellulose will be competitive with making it from corn or other food crops.
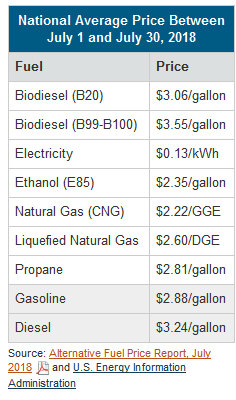
Like gasoline and diesel fuel, alternative fuel prices can vary according to market forces, politics, weather and location. With ethanol, when corn prices are high, ethanol becomes more costly and costs more than gasoline. Conversely, when corn prices are low, ethanol is less expensive and costs less than gasoline. Below is a comparison of alternative fuel prices for July 2018:

Hydrogen is one of the most abundant elements on Earth. However, it does not exist in a free state but is combined with other elements such as oxygen to form water (H2O), methane (CH4) and other hydrocarbon compounds. So it has to be separated from these other elements to create pure hydrogen. Hydrogen can be separated by water using an electric current (electrolysis). But most commercial hydrogen is produced from natural gas using a high temperature steam reforming process. This makes hydrogen about three times more expensive than natural gas.
The main advantage of hydrogen as an alternative motor fuel is that it burns clean, producing only water vapor but no carbon dioxide. However, the high temperature at which it burns can form oxides of nitrogen so combustion temperatures have to be limited by EGR, and some type of exhaust aftertreatment has to be used to reduce NOX emissions in the exhaust.
Hydrogen is a low density gaseous fuel, similar to propane or natural gas. According to the U.S. Dept. of Energy, the energy in 2.2 pounds (1 kilogram) of hydrogen gas is about the same as the energy in 1 gallon (6.2 pounds, 2.8 kilograms) of gasoline. However, because hydrogen has a low volumetric energy density, it must be stored as a highly compressed gas (5,000 to 10,000 PSI) to achieve the driving range of a conventional vehicle. This requires heavy and expensive fuel tanks to store the fuel.
Internal combustion engines are only about 30 to 35 percent efficient, so a better way to use hydrogen as an alternative fuel is to use it as a power source for an electric vehicle rather than an internal combustion engine. as the power source for a fuel cell in an electric vehicle. When used in a fuel cell, energy conversion efficiency can be 60 percent or higher. Toyota has a hydrogen fuel cell powered vehicle called the Mirai that has a range of over 400 miles on a tank of hydrogen.

AFV Alternative Fuel Vehicle
B20 Biodiesel 20 percent blend with ultra low sulfur diesel
B100 Biodiesel 100 percent from renewable sources
CNG Compressed Natural Gas
E10 10 percent blend of ethanol with 90 percent gasoline
E15 15 percent blend of ethanol with 85 percent gasoline
E85 A blend of 85% ethanol and 15% gasoline
FFV Flexible Fuel Vehicle (Ford) Can run ethanol/gasoline mixtures
GTMV Gasoline Tolerant Methanol Vehicle (Chrysler)
LPG Liquefied Petroleum Gas (propane)
LNG Liquefied Natural Gas (methane)
M85 Mixture of 85% methanol alcohol & 15% gasoline
NGV Natural Gas Vehicle (methane)
VFV Variable Fuel Vehicle (GM)
 More Alternative Fuel Articles:
More Alternative Fuel Articles: Click Here To See More Automotive Technical Articles
Click Here To See More Automotive Technical Articles